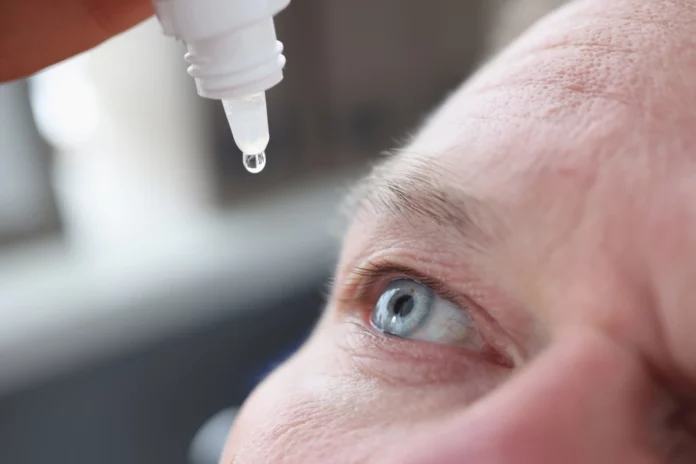
The Centers for Disease Control and Prevention (CDC) says tainted eyedrops have resulted in two more fatalities and increased instances of vision loss.
The contaminated drops, EzriCare Artificial Tears, were created by EzriCare and Delsam Phama, and were recalled in February.
The CDC is investigating the outbreak, monitoring infections and recognizing cases in 16 states nationwide.
The bacterium responsible for the epidemic, Pseudomonas aeruginosa, is resistant to standard antibiotics.
The CDC has reported a total of 68 infections, leading to three deaths and eight occurrences of vision loss.


Due to the severity of the infections, four individuals have undergone surgery to remove an eyeball.
The contaminated eyedrops were manufactured by Global Pharma Healthcare in India, where Pseudomonas aeruginosa is frequently linked with hospital outbreaks and can be transmitted via contaminated hands or medical equipment.
News by Breaking911
![]()